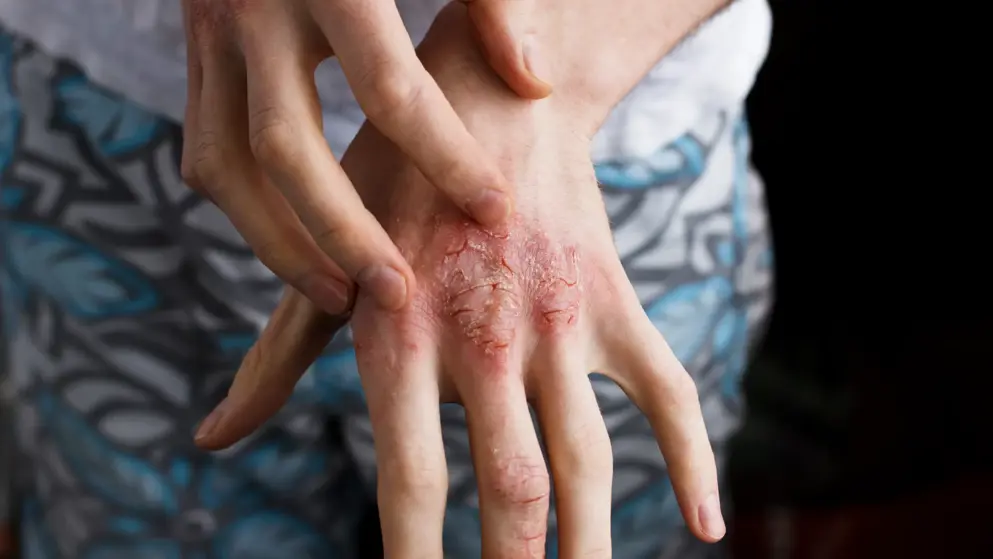
CHMP Supports Nemluvio for Atopic Dermatitis & PN
Galderma announced that the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) has adopted a positive opinion and recommended granting the marketing authorization of nemolizumab for the treatment of both atopic dermatitis and prurigo nodularis in the European Union (EU).
The CHMP has recommended nemolizumab’s approval for subcutaneous use for the treatment of moderate-to-severe atopic dermatitis in patients aged 12 years and older who are candidates for systemic therapy, and for subcutaneous use for the treatment of adults with moderate-to-severe prurigo nodularis who are candidates for systemic therapy. This follows its approval for the treatment of adults with prurigo nodularis by the United States FDA in August 2024.
This positive CHMP opinion is based on robust results from the phase III ARCADIA and OLYMPIA clinical trial programs in atopic dermatitis and prurigo nodularis, respectively.
The phase III ARCADIA 1 and 2 trials evaluated the efficacy and safety of nemolizumab in 1,728 adolescent and adult patients with moderate-to-severe atopic dermatitis. Results demonstrated that patients treated with nemolizumab, administered subcutaneously every four weeks in combination with background topical corticosteroids (TCS), with or without topical calcineurin inhibitors (TCI), showed statistically significant improvements in co-primary and key secondary endpoints, when compared to placebo in combination with TCS, with or without TCI, after 16 weeks of treatment, with significant itch relief observed as early as Week 1
The phase III OLYMPIA 1 and 2 trials evaluated the efficacy and safety of nemolizumab administered subcutaneously every four weeks in more than 500 patients with prurigo nodularis. The trials met both their primary and key secondary endpoints, demonstrating that treatment with nemolizumab resulted in significant and clinically meaningful improvements in itch and skin nodules at Week 16, with rapid reductions in itch observed as early as Week 4.Nemolizumab was well tolerated in both trials, and its safety profile was generally consistent with earlier data, and between trials